Interventional Cardiology
Occlutech PFO devices, ASD devices and Delivery Sets
Occlutech is a leading specialist provider of minimally invasive cardiac devices
Our mission is to improve the quality of life for people with heart conditions, collaborating with leading healthcare professionals to design, manufacture, and commercialize best in class cardiac devices.
Occlutech PFO Occluder

Occlutech PFO Occluder
Occlutech ASD Occluder
Occlutech ASD Occluder
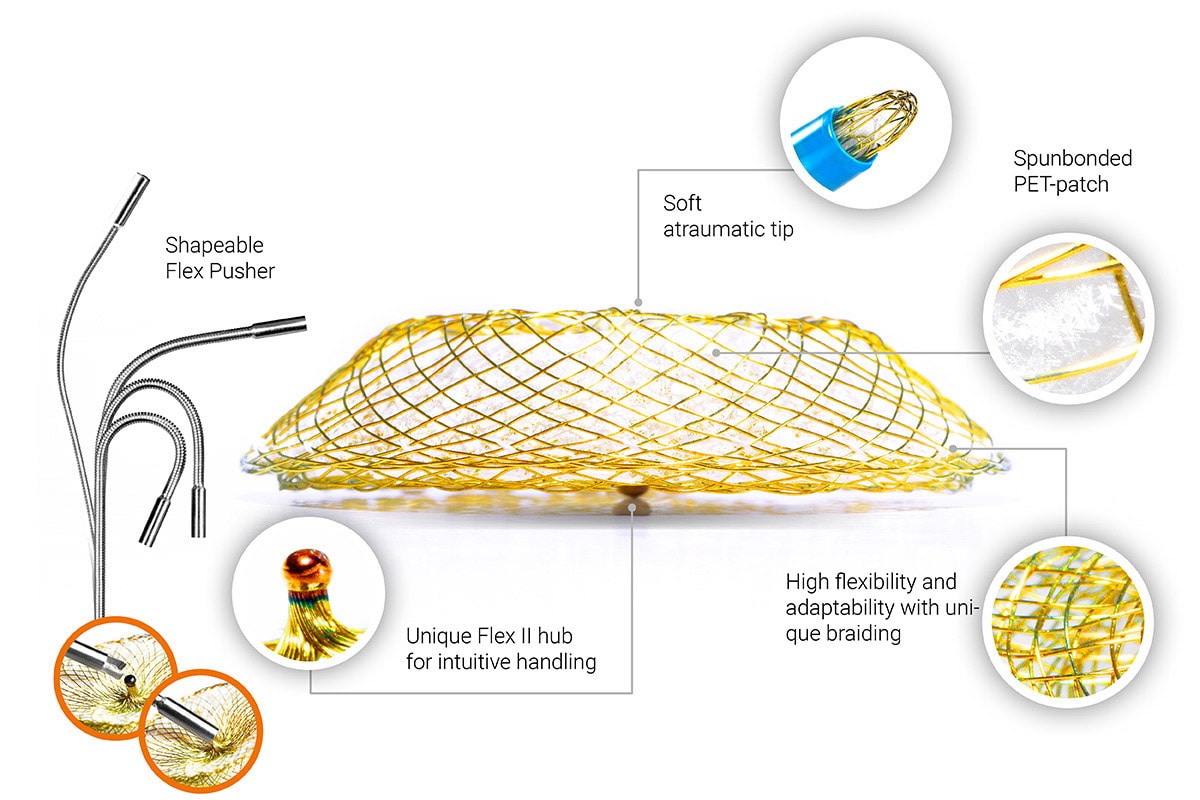
Occlutech Delivery Set
A. Dilator
The dilator (A) is made from smooth conformable low friction radio-opaque material. The dilator is inserted into the sheath (B) to form a supporting inner body. The inner lumen of the dilator has 0.035” guide wire compatibility.
B. Delivery sheath
The sheath (B) consists of a smooth outer layer and a low friction inner layer reinforced with metal braiding. The braid improves the strength of the sheath and provides radiopacity. The sheath has a proximal female connector for connection to the dilator (A) or to the loader (C).
C. Transparent loader
The loader (C) is made of low friction material and is transparent to allow inspection for air bubbles after pulling the occluder into the loader.
D. Y-connector with hemostasis valve
The Y-connector (D) is a commercially available connector with a sideport and hemostasis valve. The valve allows for leak-proof passage of the Occluder delivery wire. The side-port permits flushing before and during implantation.
Venus-P – Transcatheter Pulmonary Valve System
Product Information
A self-expanding Nitinol multilevel support frame with a tri-leaflet porcine pericardium tissue valve.
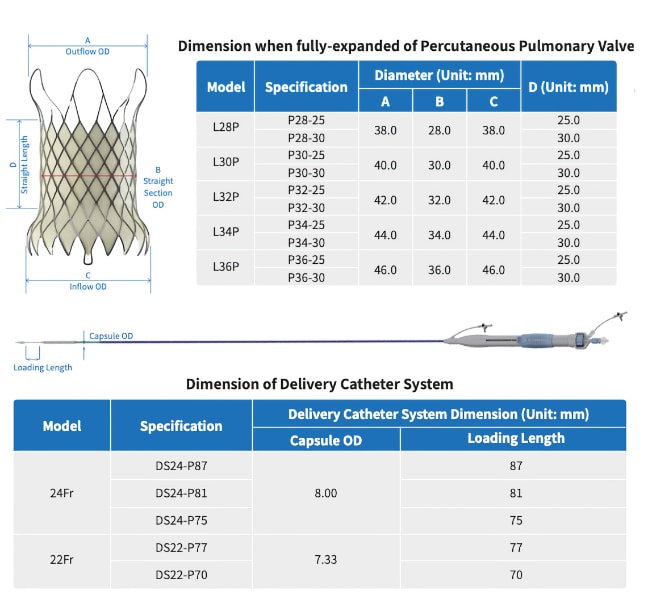
A Uni-body design – inflow, middle, outflow
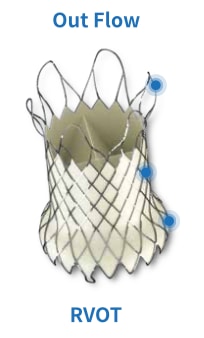
- Outflow-smooth rounded frame for ultimate artery
protection
- Middle-frame for housing the valves and expansion
in the pulmonary main. Patient special length can be designed and made
quickly
- Inflow-Conform to the RVOT and tapered at the
end for artery protection
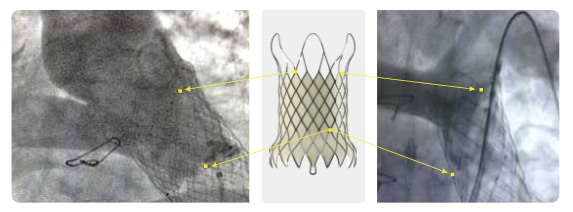
Deployment Position
-
- Flared outflow end secures anchoring at PA bifurcation
- RO markers indicate the positions of anchoring at the top and the valve location
- Uncovered end maintains L/R PA patency
- Flared and covered inflow end conform to the dilated RVOT
Texray™ Radiation Protection Textile
Texray has invented the only radiation protection textile in the world. Their unique material and product design can reduce scatter radiation towards the head and neck by up to 97% to enhance the safety of healthcare professionals. The Texray product line can be used in interventional electrophysiology, cardiology, radiology and endovascular surgery procedures where RSK Medical already offers other specialty products.

INNOVATION MEETS COMFORT & DESIGN
Personal radiation protection devices used to protect from harmful ionizing radiation are sometimes unpractical and difficult to use, resulting in unprotected body parts. Some protective garments are uncomfortable and heavy.¹ The commonly used materials do not allow a human-centered design or durable protection.
With Texray we may change that. We have invented a revolutionizing novel material platform with a woven textile technology. The essence of the textile is the attenuating capacity which protects you from potentially harmful radiation. With our innovation we enable unparalleled comfortability with pliable and breathable designs. Leaving you to stay focused, highly protected and with peace of mind in the OR.
WHY DURABILITY IS IMPORTANT
Due to the limitations of most radiation protection products, such as caps, covers and aprons must be stored hanging, to reduce folds. Folds are the sources of pinholes, which develop quickly into major tears. These tears are not always easy to feel through the surface of the material, but are hopefully detected in annual quality testing. Until then, the compromised product is a significant health and safety risk.
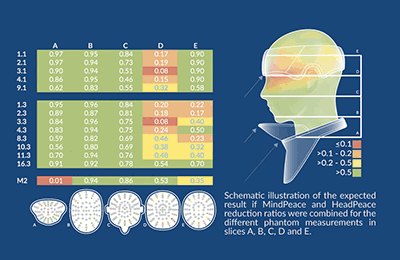
Evaluation of a novel thyroid collar designed to reduce head and neck radiation exposure during X-ray guided interventions.
Larsson M, Apell P, Jonasson P, Lundh C. Gothenburg Sweden. Poster presentation at the LINC meeting, Leipzig, Germany, 2020.
METHOD
Texray HeadPeace and MindPeace were evaluated for reducing radiation exposure to the head and neck during clinical interventions. A standard thyroid collar and a ceiling-mounted lead glass shield were used as comparisons.
RESULTS
Preliminary TLD data showed that MindPeace (extended thyroid collar) reduce radiation in the throat, chin, and ear slices (A, B, C). Some shielding effect using MindPeace was also seen in the brain and skull slices (D, E). For the standard thyroid collar a reduction was only seen in the throat slice (A) and partly in the chin slice (B). HeadPeace showed a shielding effect in the skull slice (E) up to 2 cm depth where HeadPeace covered the phantom head.
CONCLUSION
For optimal radiation protection, devices that protect the entire operators head against scattered radiation should be used. However, in clinical practice, this may not always be possible. Moreover, particularly during complex cases when radiation protection may be most relevant, optimal placement of lead glasses may not always be achievable. HeadPeace and MindPeace may therefore provide complimentary protection.